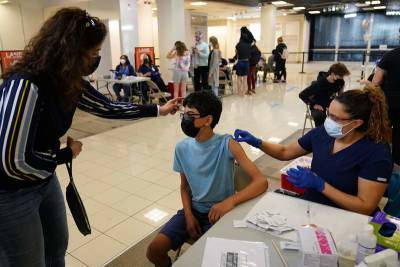
WHO approves China's Sinopharm COVID-19 vaccine for emergency use
GENEVA - The World Health Organization on Friday gave its authorization for emergency use of a COVID-19 vaccine manufactured by China's Sinopharm, potentially paving the way for millions of the doses to reach needy countries through a U.N.-backed program rolling out coronavirus vaccines.The decision by a WHO technical advisory group opens the possibility that the Sinopharm vaccine could be included into the U.N.-backed COVAX program in coming weeks or months, and distributed through UNICEF and WHO’s regional office in the Americas.Sinopharm has released very little data publicly, aside from efficacy numbers for its two vaccine shots – one developed by its Beijing Institute of Biological Products and the other by the Wuhan Institute of.