Moderna, J&J COVID-19 booster shots: FDA panel meets to review data
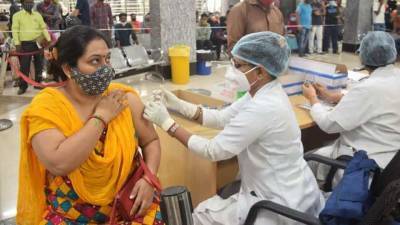
WASHINGTON - A panel of independent advisers to the U.S. Food and Drug Administration will convene on Thursday and Friday to review data and vote on whether the millions of Americans who received the Moderna or Johnson & Johnson COVID-19 vaccine should get a booster shot.
The FDA’s Vaccines and Related Biological Products Advisory Committee, scheduled to meet both days from 8:30 a.m. to 5 p.m. ET, is the first step in a review process of deciding whether the extra doses should be dispensed, and if so, who should get them and when.
Experts will discuss whether a third shot of Moderna’s two-dose vaccine should contain just half the original dose and what’s the best timing for a second shot of the single-dose J&J vaccine.